What is Temperature Mapping in Manufacturing Industry?
- Jan 28
- 3 min read
Temperature Mapping is a scientific and risk-based study used to understand and control temperature distribution within a defined space such as warehouses, cold rooms, refrigerators, freezers, cleanrooms, manufacturing areas, and transport systems.
In pharma, biotech, and food industries, many materials are temperature sensitive and their quality, safety, and efficacy depend on being stored and handled within approved temperature limits. Temperature mapping helps verify that every part of the area consistently maintains these limits under normal operating conditions, including worst-case scenarios like maximum load, minimum load, seasonal variation, and power interruptions.
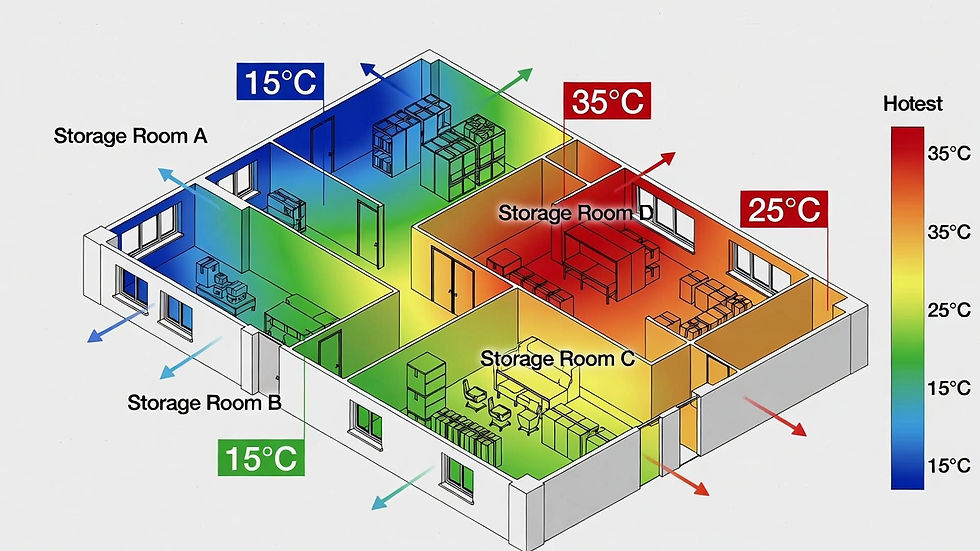
typical temperature layout of a storage/ warehouse
The study uses calibrated data loggers placed at strategic locations to record temperature over a defined period. The data is analyzed to identify hot spots, cold spots, and areas of fluctuation. These results are then used to define monitoring sensor locations, alarm limits, storage layouts, and corrective actions, ensuring regulatory compliance and long-term control of the environment.
Example of Temperature Mapping
Consider a medicine that must be stored between 10°C and 18°C to maintain its safety, potency, and effectiveness. At a pharmacy or small medical store, this requirement is easy to manage because the product is stored in a refrigerator with uniform cooling.
However, the same approach cannot be applied to a large pharmaceutical warehouse. Warehouses are much larger, have varying airflows, multiple doors, racking systems, and temperature gradients. A single thermostat or cooling unit cannot guarantee uniform temperature throughout the space.
This is where Temperature Mapping becomes essential.
During temperature mapping, multiple calibrated sensors are placed across the warehouse—at different heights, corners, near doors, and inside racks. Data is collected over defined conditions (summer, winter, loaded, empty, door openings) to identify hot spots and cold spots.
The study proves whether the warehouse can consistently maintain the required temperature across all locations, not just at concentrated points. Based on the results, manufacturers can:
Optimize cooling systems
Define monitoring sensor locations
Establish alarm limits
Ensure compliance with GMP and regulatory expectations
Temperature mapping ensures that every unit of medicine, regardless of where it is stored in the warehouse, remains within the approved temperature range and retains its quality throughout storage.

storage temperature monitoring
Manual Temperature Mapping
Manual temperature mapping is typically used for one-time qualification studies such as initial validation, requalification, or after major changes. In this approach:
Standalone calibrated data loggers are placed at predefined locations
Sensors record temperature at set intervals for a defined duration (e.g., 7–15 days)
Data is downloaded manually at the end of the study
Analysis is performed to identify hot and cold spots
A report is generated to demonstrate compliance
This method is cost-effective and suitable for small warehouses, refrigerators, incubators, transport boxes, or when mapping is required only periodically.
Automated Temperature Mapping
Automated mapping uses a continuous monitoring system with fixed sensors connected to a central software platform. In this approach:
Sensors continuously capture temperature data in real time
Data is stored automatically and securely
Alarms are triggered if limits are breached
Trends and deviations can be analyzed instantly
Audit trails and reports are generated automatically
Automated systems are preferred for large warehouses, GMP manufacturing areas, cold rooms, and high-risk storage zones, where continuous control is required. They also support 21 CFR Part 11, data integrity, and regulatory inspections.
Manual mapping is used to prove the system works, while automated mapping is used to continuously verify that it keeps working. In GMP environments, both are often used together: manual mapping for qualification and automated monitoring for routine control.
Read More about our Temperature Mapping service. Contact Now

